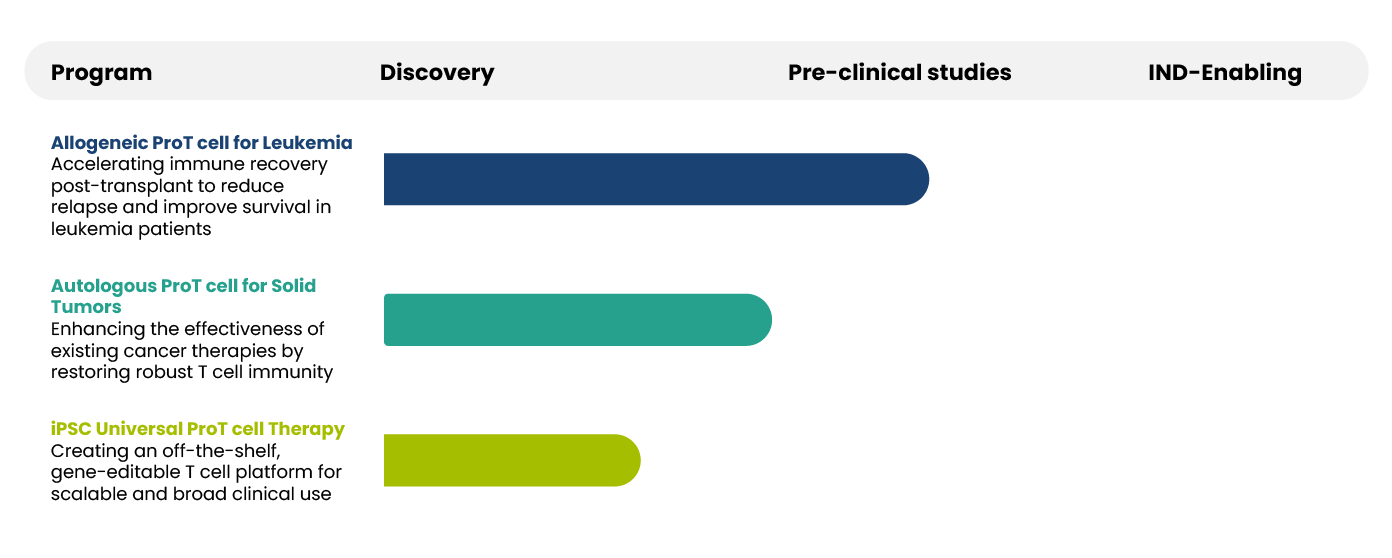
ProTgen Pipeline
1st Gen:
Allogeneic HSCT + ProT cell infusion for leukemia
Allogeneic HSCT for adult leukemia patients remains the best option for many
T cell recovery at 3 months correlates with ⬆️ overall survival, ⬇️ decreased relapse
ProT cell therapy takes 2 weeks post-HSCT (same as allo-CD34+ HSC donation)
T cell recovery expected ~3 months or less, making for practical dose selection PoC
ProTgen’s Platform Development Strategy
Clinic ready by 2027
2nd Gen:
Autologous ProT cell infusion for solid tumors
Standard of care includes radiation, ADC, oncolytic virus, CAR-Ts, etc.
ProT cell therapy likely to improve effectiveness of bispecific T-cell engagers (e.g., CD3-DLL3) and/or neo antigen vaccine (e.g., mRNA) treatments
Cells can be edited to include transient growth factors or CAR
3rd Gen:
Universal iPSC-derived ProT cells: scalable off-the-shelf immunotherapy
Hypo-immune iPSC-derived
Editable, cryopreservable
Expand in vivo after administration
Source of non-exhausted, expanding T cell repertoire

First Generation Platform:
Allogeneic ProT Cells for HSCT
Clinical Concepts for 2nd and 3rd Generation ProT Cells: Autologous and iPSC Off-the-Shelf
Broad T cell diversity improves survival in many solid tumor cancers (list expanding):
NSCLC
Metastatic breast cancer
Advanced bladder cancer
Advanced melanoma treated with anti-CTLA4 or anti-PD1
Pancreatic cancer treated with anti-CTLA4